A) I,III
B) I,IV
C) II,III
D) II,IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following structures I-IV.Which two species represent resonance structures? 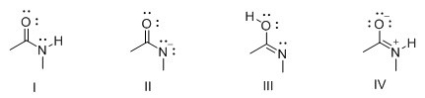
A) I and II
B) I and III
C) I and IV
D) II and IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about Brønsted-Lowry acids and bases is true?
A) Loss of a proton from a base forms its conjugate acid.
B) Loss of a proton from an acid forms its conjugate base.
C) Gain of a proton by an acid forms its conjugate base.
D) Brønsted-Lowry acid-base reactions always result in the transfer of a proton from a base to an acid.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is not a Lewis acid?
A) AlCl3
B) HCl
C) H2O
D) CBr4
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following structures I-IV.Which two species represent a conjugate acid-base pair? 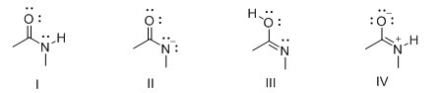
A) I and II
B) I and III
C) I and IV
D) II and III
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would have the lowest pKa?
A) ClCH2CH2CH2CH2COOH
B) CH3CHClCH2CH2COOH
C) CH3CH2CHClCH2COOH
D) CH3CH2CH2CHClCOOH
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following species is the conjugate acid of ammonia,NH3?
A) H4N
B) H3N+
C) H2N-
D) H4N+
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the electrophilic site in the following compounds? 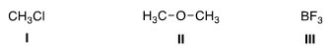
A) I = Carbon; II = carbon; III = boron.
B) I = Chlorine; II = carbon; III = boron.
C) I = Carbon; II = oxygen; III = boron.
D) I = Carbon; II = carbon; III = fluorine.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following species can be both Lewis acid and Lewis base? 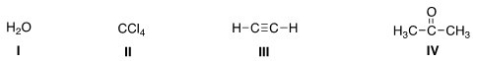
A) I,III,IV
B) I,II,IV
C) II,III,IV
D) I,II,III
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of increasing acidity,putting the least acidic first. 
A) III < I < IV < II
B) III < IV < I < II
C) II < I < IV < III
D) III < I < II < IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following species is the strongest base?
A) HO-
B) H2N-
C) CH3COO-
D) Cl-
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of decreasing acidity,putting the most acidic first. 
A) IV > II > III > I
B) III > II > IV > I
C) I > II > IV > III
D) III > IV > II > I
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is a correct definition for a Brønsted-Lowry acid?
A) Proton acceptor
B) Electron pair donor
C) Electron pair acceptor
D) Proton donor
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following species is the conjugate base of methanol,CH3OH?
A) CH3OH2+
B) CH3O-
C) CH3-
D) CH4
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of decreasing acidity,putting the most acidic first. 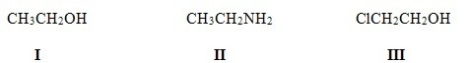
A) I > II > III
B) III > II > I
C) II > III > I
D) III > I > II
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about a Brønsted-Lowry base is true?
A) The net charge may be zero,positive,or negative.
B) All Brønsted-Lowry bases contain a lone pair of electrons or a π bond.
C) All Brønsted-Lowry bases contain a proton.
D) The net charge may be zero or positive.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ranks the compounds in order of increasing basicity,putting the least basic first?
A) CH3NH2 < CH3OH < CH4
B) CH3OH < CH3NH2 < CH4
C) CH4 < CH3NH2 < CH3OH
D) CH4 < CH3OH < CH3NH2
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the conjugate acid in the following reaction? 
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements explain why HBr is a stronger acid than HF?
A) Br- is more stable than F- because Br- is larger than F-.
B) Br- is less stable than F- because Br- is larger than F-.
C) Br- is more stable than F- because Br- is less electronegative than F-.
D) Br- is less stable than F- because Br- is less electronegative than F-.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 59 of 59
Related Exams