A) Only I
B) Only II
C) Only III
D) Only I and II
F) A) and C)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 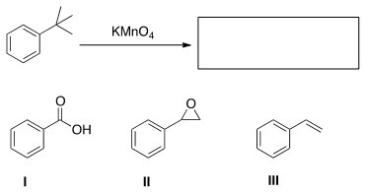
A) I
B) II
C) III
D) None of these
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major product of the following reaction? 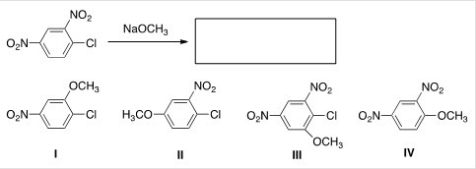
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is (are) the product(s) of the following reaction? 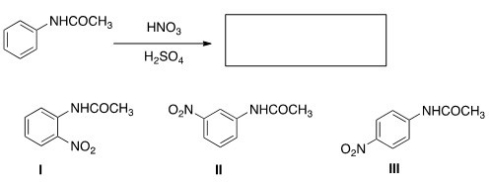
A) Only I
B) Only II
C) Only III
D) Only I and III
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which aryl fluoride reacts the fastest with NaOH? 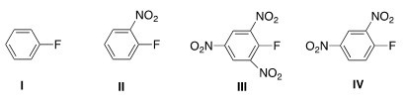
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following monobromonation reaction? 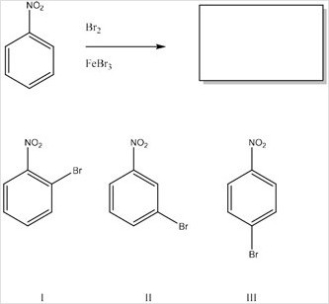
A) Only I
B) Only II
C) Only III
D) Only I and III
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is sulfuric acid used in aromatic nitration?
A) To keep the reaction from getting too basic
B) To form the active electrophile NO2+
C) To protonate the aromatic ring
D) To keep the reaction from getting too acidic
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following sequence of reactions? 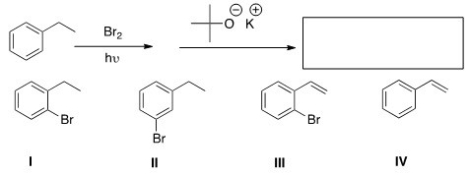
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which set of reagents would most likely bring about the following transformation? 
A) Br2 and FeBr3
B) NBS and light
C) Br2 in CCl4
D) NaBr and H2O
F) A) and B)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Which of the following halides will not work as an electrophile in a Friedel-Crafts alkylation reaction? 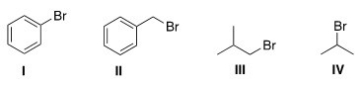
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following activating groups in order of decreasing strength of activation,listing the most activating first. 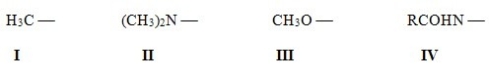
A) IV > II > III > I
B) II > III > IV > I
C) III > IV > II > I
D) II > IV > III > I
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product obtained from the following reaction? 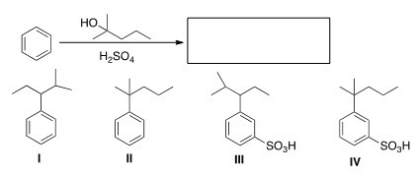
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product obtained from the following reaction? 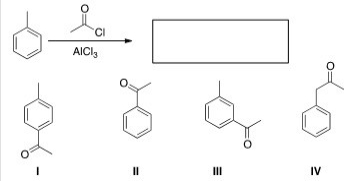
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How can polyalkylation be minimized in Friedel-Crafts alkylation?
A) Use a large excess of alkyl halide relative to the aromatic compound.
B) Use a large excess of benzene relative to the alkyl halide.
C) Use an alkyl halide without a Lewis acid catalyst.
D) Use a large excess of the Lewis acid catalyst.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product obtained from the following reaction? 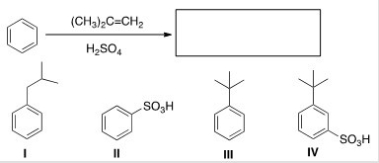
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following substituents are deactivators in electrophilic aromatic substitution?
A) HO ¾
B) CH3NH ¾
C) CH3O ¾
D) (CH3) 3N+¾
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What are the product(s) of the following reaction? 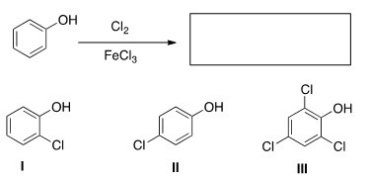
A) Only I
B) Only II
C) Only I and II
D) Only III
F) All of the above
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
What is the major organic product obtained from the following reaction? 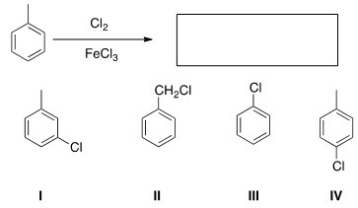
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 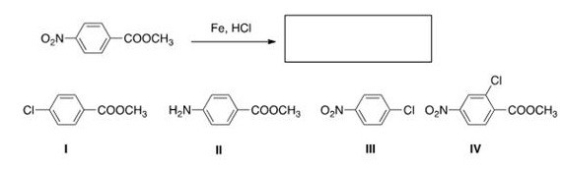
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is the nitro group a meta director?
A) Because it is sterically very large
B) Because it adds electron density to the meta position,thus activating it
C) Because it stabilizes the intermediate cation
D) Because it removes more electron density from the ortho and para positions than the meta position,thus deactivating the meta position less
F) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 60
Related Exams