B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Valence bond theory predicts that carbon will use _____ hybrid orbitals in the carbonate anion, CO3-.
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The nitrosonium ion, NO+, forms a number of interesting complexes with nickel, cobalt, and iron. According to molecular orbital theory, which of the following statements about NO+ is correct?
A) NO+ has a bond order of 2 and is paramagnetic.
B) NO+ has a bond order of 2 and is diamagnetic.
C) NO+ has a bond order of 3 and is paramagnetic.
D) NO+ has a bond order of 3 and is diamagnetic.
E) None of these choices are correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following statements about orbital hybridization is incorrect?
A) The carbon atom in CH4 is sp3 hybridized.
B) The carbon atom in CO2 is sp hybridized.
C) The nitrogen atom in NH3 is sp2 hybridized.
D) sp2 hybrid orbitals are coplanar, and at 120° to each other.
E) sp hybrid orbitals lie at 180° to each other.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In molecular orbital theory, combination of two 2p atomic orbitals may give rise to either σ or π type molecular orbitals.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Valence bond theory predicts that xenon will use _____ hybrid orbitals in XeOF4.
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
According to valence bond theory, overlap of bonding orbitals of atoms will weaken a bond, due to electron-electron repulsion.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Hybrid orbitals of the sp3d type occur in sets of four.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A molecule with the formula AX3E uses _________ to form its bonds.
A) s and p atomic orbitals
B) sp3 hybrid orbitals
C) sp2 hybrid orbitals
D) sp hybrid orbitals
E) sp3d2 hybrid orbitals
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to valence bond theory, which orbitals on N and H overlap in the NH3 molecule?
A) 2p on N overlaps with 2s on H
B) 2p on N overlaps with 1s on H
C) 2s on N overlaps with 1s on H
D) sp3 on N overlaps with sp on H
E) sp3 on N overlaps with 1s on H
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Carbon uses ______ hybrid orbitals in ClCN.
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In the valence bond treatment, a 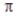 bond is formed when two 2p orbitals overlap side to side.
bond is formed when two 2p orbitals overlap side to side.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many sigma and pi bonds, respectively, are in the molecule below? CH3CH2CHCHCH3?
A) 16, 1
B) 16, 0
C) 15, 1
D) 15, 0
E) 14, 1
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
A carbon-carbon triple bond in a molecule may give rise to the existence of cis and trans isomers.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the shape (geometry) of PCl3 and then decide on the appropriate hybridization of phosphorus in this molecule. (Phosphorus is the central atom.)
A) sp3
B) sp2
C) sp
D) sp3d
E) sp3d2
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to valence bond theory, the triple bond in ethyne (acetylene, C2H2) consists of
A) three σ bonds and no ![]() bonds.
bonds.
B) two σ bonds and one ![]() bond.
bond.
C) one σ bond and two ![]() bonds.
bonds.
D) no σ bonds and three ![]() bonds.
bonds.
E) None of these choices are correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to molecular orbital (MO) theory, the twelve outermost electrons in the O2 molecule are distributed as follows:
A) 12 in bonding MOs, 0 in antibonding MOs.
B) 10 in bonding MOs, 2 in antibonding MOs.
C) 9 in bonding MOs, 3 in antibonding MOs.
D) 8 in bonding MOs, 4 in antibonding MOs.
E) 7 in bonding MOs, 5 in antibonding MOs.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Overlap of two sp2 hybrid orbitals produces a 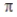 bond.
bond.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Valence bond theory predicts that bromine will use _____ hybrid orbitals in BrF3.
A) sp2
B) sp3
C) sp3d
D) sp4
E) sp3d2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In molecular orbital theory, combination of two atomic orbitals produces two molecular orbitals.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 48
Related Exams