A) 1.49 M
B) 1.66 M
C) 7.82 M
D) 12.5 M
E) 16.6 M
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following changes will NOT affect the total pressure of gas in a container, assuming all other factors remain constant?
A) The frequency of collisions of molecules with the walls is increased.
B) The average velocity of the molecules is lowered.
C) The temperature of the sample is altered.
D) Half of the molecules are replaced by an equal number of molecules of a gas with a different molecular weight.
E) The total number of molecules is altered.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Nitrogen dioxide is a red-brown gas that is responsible for the color of photochemical smog. A sample of nitrogen dioxide has a volume of 28.6 L at 45.3 C and 89.9 kPa. What is its volume at STP?
A) 21.8 L
B) 27.6 L
C) 29.6 L
D) 37.6 L
E) 153 L
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pressure in a 7.50-L flask if 0.15 mol of carbon dioxide is added to 0.33 mol of oxygen? The temperature of the mixture is 48.0 C.
A) 0.252 atm
B) 0.592 atm
C) 1.69 atm
D) 3.96 atm
E) 4.80 atm
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Helium gas is being pumped into a rigid container at a constant temperature. As a result, the pressure of helium in the container is increasing. Select the one correct statement below.
A) As the pressure increases, helium atoms move faster, on average.
B) As the pressure increases, helium atoms move more slowly, on average.
C) As the pressure increases, the volume of the container must decrease.
D) As the pressure increases, helium atoms stay closer to the wall of the container, on average.
E) As the pressure increases, there are more collisions of helium atoms with the container wall.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At very high pressures (~ 1000 atm) , the measured pressure exerted by real gases is greater than that predicted by the ideal gas equation. This is mainly because
A) such high pressures cannot be accurately measured.
B) real gases will condense to form liquids at 1000 atm pressure.
C) gas phase collisions prevent molecules from colliding with the walls of the container.
D) of attractive intermolecular forces between gas molecules.
E) the volume occupied by the gas molecules themselves becomes significant.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the rms speed of carbon dioxide molecules at STP.
A) 12.4 m/s
B) 155m/s
C) 393 m/s
D) 1.55 * 105 m/s
E) The answer can't be calculated without more data.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
For real gases, PV > nRT, always.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
"The volume of an ideal gas is directly proportional to its absolute temperature at constant pressure and number of moles" is a statement of ________________ Law.
A) Charles's
B) Boyle's
C) Amontons's
D) Avogadro's
E) Dalton's
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the statement which does NOT apply to an ideal gas.
A) There are no attractive forces between the gas molecules.
B) There are strong repulsive forces between the gas molecules.
C) The volume occupied by the molecules is negligible compared to the container volume.
D) The gas behaves according to the ideal gas equation.
E) The average kinetic energy of the molecules is proportional to the absolute temperature.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 250.0-mL sample of ammonia, NH3(g) , exerts a pressure of 833 torr at 42.4 C. What mass of ammonia is in the container?
A) 0.0787 g
B) 0.180 g
C) 8.04 g
D) 17.0 g
E) 59.8 g
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At moderate pressures (~ 200 atm) , the measured pressure exerted by CO2 gas is less than that predicted by the ideal gas equation. This is mainly because
A) such high pressures cannot be accurately measured.
B) CO2 will condense to a liquid at 200 atm pressure.
C) gas phase collisions prevent CO2 molecules from colliding with the walls of the container.
D) of attractive intermolecular forces between CO2 molecules.
E) the volume occupied by the CO2 molecules themselves becomes significant.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A ballerina weighs 103 lbs and is up on her toes with only 10.0 cm2 of her slippers in contact with the floor. What pressure is she exerting on the floor?
A) 4.59 *105 Pa
B) 4.59 * 103 Pa
C) 4.59 * 101 Pa
D) 4.59 * 10¯1 Pa
E) 4.59 * 10¯3 Pa
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
According to the kinetic theory of gases, in a collision between two molecules the kinetic energy of one molecule will decrease by the same amount that the kinetic energy of the other one increases.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A compressed gas cylinder containing 1.50 mol methane has a volume of 3.30 L. What pressure does the methane exert on the walls of the cylinder if its temperature is 25 C?
A) 9.00 *¯2 atm
B) 0.933 atm
C) 1.11 atm
D) 1.70 atm
E) 11.1 atm
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
For a pure gas sample, the average kinetic energy is also the most probable kinetic energy.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Lithium oxide is an effective absorber of carbon dioxide and can be used to purify air in confined areas such as space vehicles. What volume of carbon dioxide can be absorbed by 1.00 kg of lithium oxide at 25 C and 1.00 atm? Li2O(aq) + CO2(g) Li2CO3(s)
A) 687 mL
B) 819 mL
C) 687 L
D) 819 L
E) 22.4 L
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the lines on the figure below is the best representation of the relationship between the volume of a gas and its absolute temperature, other factors remaining constant? 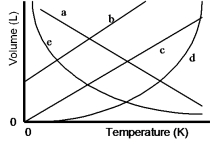
A) a
B) b
C) c
D) d
E) e
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrogen gas exerts a pressure of 466 torr in a container. What is this pressure in atmospheres?
A) 0.217 atm
B) 0.466 atm
C) 0.613 atm
D) 1.63 atm
E) 4.60 atm
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A gas mixture, with a total pressure of 300. torr, consists of equal masses of Ne (atomic weight 20.) and Ar (atomic weight 40.) . What is the partial pressure of Ar, in torr?
A) 75 torr
B) 100. torr
C) 150. torr
D) 200. torr
E) None of these choices is correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 103
Related Exams