A) [1] LiAlH4, [2] H2O
B) [1] CH2=CHLi, [2] H2O
C) [1] (CH2=CH) 2CuLi, [2] H2O
D) [1] DIBAL-H, [2] H2O
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction?
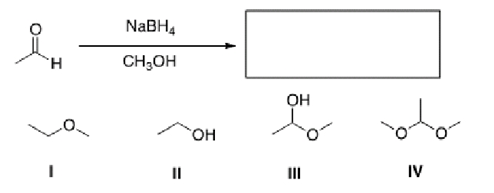
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What will be the product of the following reaction (before any aqueous work-up) ?
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
What reagent would be used to reduce an amide to an amine?
A) NaBH4
B) LiAlH(OtBu) 3
C) LiAlH4
D) FeCl3
F) C) and D)
Correct Answer

verified
Correct Answer
verified
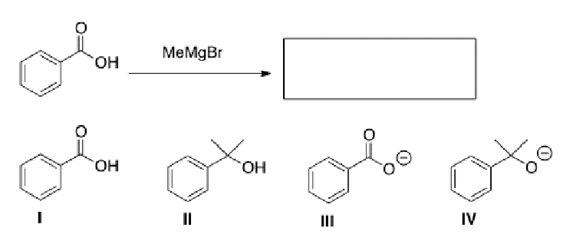
Multiple Choice
What is the product of the following reaction?
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A carbonyl group, C=O, and an alkene, C=C, double bonds are both sp2 hybridized.However, the chemistry of These two functional groups is very different.This can be explained by which of the following statements?
A) The bond angle of the carbonyl is larger than the bond angle of the alkene.
B) The electronegative oxygen of the C=O group makes this bond polar.
C) The bond of the C=C is longer that the bond of the C=O.
D) There is more steric crowding in the carbonyl than in the alkene.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction?
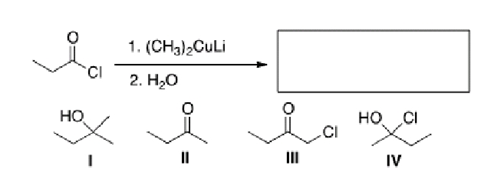
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction?
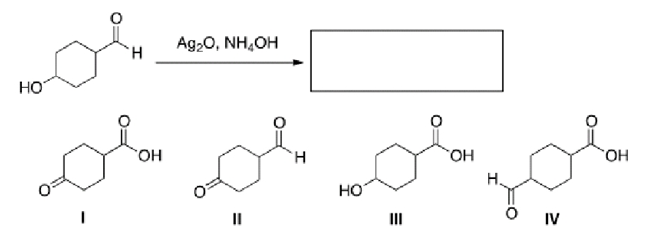
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction?
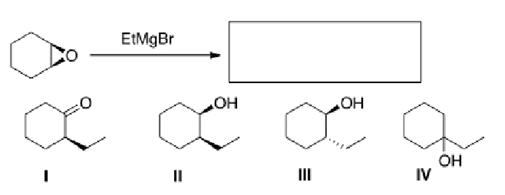
A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the missing reagent in the reaction below?
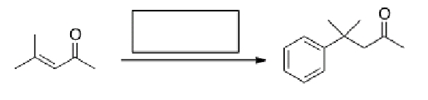
A) PhMgBr then H2O
B) Benzene, AlCl3
C) Ph2CuLi then H2O
D) PhLi then H2O
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Both LiAlH4 and NaBH4 are reducing agents.Which statement about these reagents is true?
A) Both reagents contain polar metal-hydrogen bonds.The polarity of the B-H bond is greater than the polarity of the Al-H bond, so LiAlH4 is the stronger reducing agent.
B) Both reagents contain polar metal-hydrogen bonds.The polarity of the B-H bond is greater than the polarity of the Al-H bond, so LiAlH4 is the weaker reducing agent.
C) Both reagents contain polar metal-hydrogen bonds.The polarity of the B-H bond is less than the polarity of the Al-H bond, so LiAlH4 is the stronger reducing agent.
D) Both reagents contain polar metal-hydrogen bonds.The polarity of the B-H bond is less than the polarity of the Al-H bond, so LiAlH4 is the weaker reducing agent.
F) All of the above
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
What is the missing reagent in the reaction below?
![What is the missing reagent in the reaction below? A) [1] LiAlH<sub>4</sub>, [2] H<sub>2</sub>O B) [1] CH<sub>3</sub>MgBr (excess) , [2] H<sub>2</sub>O C) [1] (CH<sub>3</sub>) <sub>2</sub>CuLi (excess) , [2] H<sub>2</sub>O D) [1] DIBAL-H, [2] H<sub>2</sub>O](https://d2lvgg3v3hfg70.cloudfront.net/TB5872/11ec712e_8b45_1f77_8ed2_dbb60504d577_TB5872_00.jpg)
A) [1] LiAlH4, [2] H2O
B) [1] CH3MgBr (excess) , [2] H2O
C) [1] (CH3) 2CuLi (excess) , [2] H2O
D) [1] DIBAL-H, [2] H2O
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction?
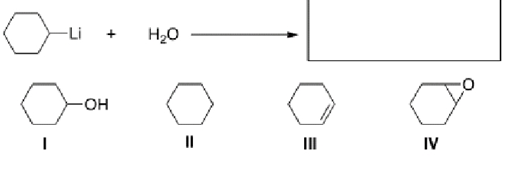
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
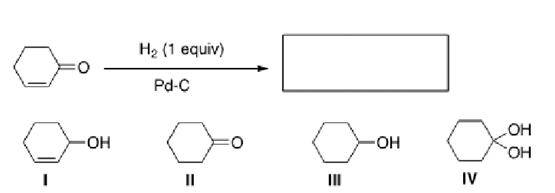
Multiple Choice
Which reagent can be used to reduce the alkene in cyclopentenone?
A) NaBH4
B) LiAlH4
C) H2 and Pd-C
D) DIBAL-H
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name of the general reaction type that aldehydes and ketones undergo?
A) Electrophilic addition
B) Nucleophilic addition
C) Nucleophilic substitution
D) Electrophilic substitution
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the starting material in the reaction below?
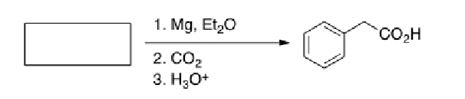
A) Bromobenzene
B) Benzyl bromide
C) Benzoic acid
D) Lithium benzoate
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction?
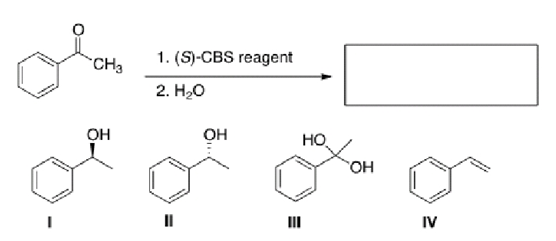
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why are ketones less reactive than aldehydes?
A) Ketones are more sterically hindered.
B) Ketones are less electron deficient due to donation from the two alkyl groups.
C) The statement is false; ketones are more reactive than aldehydes.
D) Both (a) Ketones are more sterically hindered and (b) Ketones are less electron deficient due to donation from the two alkyl groups.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why would the alcohol in the following compound need to be protected before reaction?
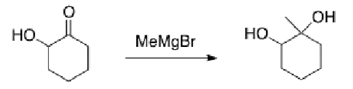
A) If it isn't protected, the product will be a carboxylic acid.
B) The Grignard reagent will react with the alcohol before the ketone.
C) Magnesium is Lewis acidic and will coordinate with the alcohol.
D) There is no need to protect the alcohol.
F) B) and D)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
What is the product of the following reaction?
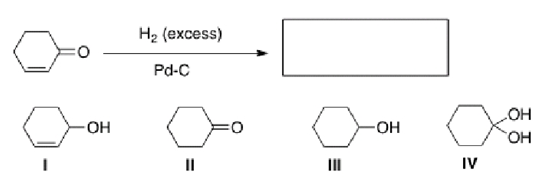
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 29
Related Exams