A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following period 3 chlorides would be expected to have the highest melting point?
A) MgCl 2
B) AlCl 3
C) SiCl 4
D) PCl 3
E) SCl 2
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following contains covalent bonds?
A) BaO
B) IBr
C) Mg
D) LiBr
E) Cu
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The lattice energy is the energy released when separated ions in the gas phase combine to form ionic molecules in the gas phase.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrogenation of double and triple bonds is an important industrial process. Calculate (in kJ) the standard enthalpy change ΔH° for the hydrogenation of ethyne (acetylene) to ethane. H-C≡C-H(g) + 2H2(g) → H3C-CH3(g)
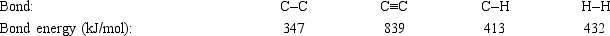
A) −296 kJ
B) −51 kJ
C) 51 kJ
D) 296 kJ
E) 381 kJ
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When two atoms form a covalently-bonded diatomic molecule, the distance between the nuclei at which the potential energy is at a minimum is called
A) the bond energy.
B) the bond length.
C) the molecular diameter.
D) the covalent radius.
E) the covalent diameter.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Combustion of a fat will release more energy than combustion of an equal mass of carbohydrate because
A) fats contain more bonds to oxygen than carbohydrates.
B) fats contain fewer bonds to oxygen than carbohydrates.
C) the total energy of the carbon-carbon and carbon-hydrogen bonds in fats is greater than the energy content of the carbon-oxygen and oxygen-hydrogen bonds in the reaction products (carbon dioxide and water) .
D) the total energy of the carbon-carbon and carbon-hydrogen bonds in fats is greater than the energy content of the bonds in carbohydrates.
E) fats have higher molar masses than carbohydrates.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange calcium, rubidium, sulfur, and arsenic in order of decreasing electronegativity.
A) S > As > Rb > Ca
B) S > As > Ca > Rb
C) As > S > Rb > Ca
D) As > S > Ca > Rb
E) None of these choices are correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Quartz (SiO2) is a solid with a melting point of 1550 °C. The bonding in quartz is best described as
A) lattice energy.
B) network attractions.
C) ionic bonding.
D) covalent bonding.
E) metallic bonding.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The majority of elements are good electrical conductors when in solid form.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Analysis of an unknown substance showed that it has a high boiling point and is brittle. It is an insulator as a solid but conducts electricity when melted. Which of the following substances would have those characteristics?
A) HCl
B) Al
C) KBr
D) SiF 4
E) I 2
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Electronegativities on Pauling's scale are calculated from ionization energies and electron affinities.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The melting points of metals are only moderately high because
A) metallic bonding is weak.
B) metals have fewer bonding electrons than nonmetals.
C) metals also have relatively low boiling points.
D) the melting process does not break the metallic bonds.
E) metals prefer to be bonded to nonmetals.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements is the most electronegative?
A) S
B) Ru
C) Si
D) Te
E) Cs
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the element whose Lewis symbol is correct.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which of the following processes does the enthalpy change (ΔH) directly represent the magnitude of the lattice energy of KCl(s) ?
A) Cl 2(g) + 2K(s) → 2KC (s)
B) KCl(s) → K +(aq) + Cl − (aq)
C) KCl(s) → K +(g) + Cl −(g)
D) KCl(s) → K(s) + Cl −(g)
E) KCl(s) → K(s) + Cl(g)
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The diameter of a chloride ion is 362 pm, and the diameter of a potassium ion is 276 pm. What is the distance between the nuclei of adjacent chloride and potassium ions in solid potassium chloride?
A) 1276 pm
B) 638 pm
C) 319 pm
D) 181 pm
E) 138 pm
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The lattice energy of MgCl2 is the energy change for which one of the following processes?
A) Mg( s) + Cl 2( g) → MgCl 2( s)
B) Mg( g) + 2Cl( g) → MgCl 2( s)
C) Mg 2+( s) + 2Cl −( g) → MgCl 2( g)
D) Mg 2+( g) + 2Cl −( g) → MgCl 2( s)
E) MgCl 2( aq) → MgCl 2( s)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following properties is least characteristic of typical ionic compounds?
A) High melting point
B) High boiling point
C) Brittleness
D) Poor electrical conductor when solid
E) Poor electrical conductor when molten
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct formula for a compound formed from barium and nitrogen.
A) BaN
B) BaN 2
C) Ba 2N 3
D) Ba 2N
E) Ba 3N 2
G) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 61
Related Exams