A) D2O
B) H2
C) NO2
D) F2
E) He
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Of the three oxides SiO2, MgO, and P4O10, the most acidic oxide is P4O10.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these choices is the electron configuration for the chloride ion?
A) [Ne]3s23p5
B) [Ne]3s23p7
C) [Ne]3s23p4
D) [Ar]4s1
E) [Ar]
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Carbon dioxide is used in all of the following ways except
A) to provide the "bite" in soft drinks.
B) in fire extinguishers.
C) as a refrigerant.
D) in the Frasch process.
E) in the manufacture of soda ash.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these compounds does not follow the octet rule?
A) PF5
B) HF
C) NF3
D) AsCl3
E) CF4
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
Sulfuric acid is produced when sulfur dioxide dissolves in water.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What group of the periodic table is the element with the electron configuration [Ar]4s23d104p2?
A) 4A
B) 3A
C) 5A
D) 2A
E) 6A
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these elements is most likely to exhibit an expanded octet in its compounds?
A) C
B) O
C) N
D) S
E) Na
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which best represents the allotrope(s) of oxygen?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below is a representation of a sample of gas with molecules having the formula XO2. Which element could NOT be represented by X? 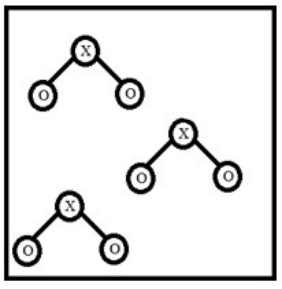
A) N
B) S
C) C
D) O
E) Cl
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of these species exists as a diatomic element at room temperature?
A) Na
B) N
C) B
D) Ar
E) P
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the strongest acid?
A) HClO3
B) HBrO3
C) HIO3
D) HIO2
E) HClO
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the charge on the monatomic ion of nitrogen, the nitride ion?
A) -3
B) +2
C) -1
D) -2
E) +1
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What type of bond is the O-H bond in CH3OH?
A) Nonpolar covalent
B) Ionic
C) Coordinate covalent
D) Polar ionic
E) Polar covalent
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these pairs consists of isoelectronic species?
A) Zn2+ and Cu2+
B) Cl- and S
C) Na+ and K+
D) Mn2+ and Ar
E) K+ and Cl-
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these elements is a liquid at room temperature?
A) Hydrogen
B) Nitrogen
C) Bromine
D) Sulfur
E) Phosphorus
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Nonmetals are more electropositive than metals.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many resonance structures does the sulfur dioxide molecule have?
A) 0
B) 3
C) 1
D) 2
E) 4
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these compounds is most likely to be covalent?
A) CaO
B) CsOH
C) LiF
D) Sr(NO3) 2
E) NF3
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
What has metal and nonmetal characteristics?
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 121
Related Exams