A) 44 kJ/mol
B) 440 J/K
C) 117.9 J/K
D) -41 kJ/K
E) -440 kJ/K
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many types of rotations does a diatomic molecule exhibit?
A) 0
B) 1
C) 2
D) 3
E) 4
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below are energy level diagrams for two different atoms. 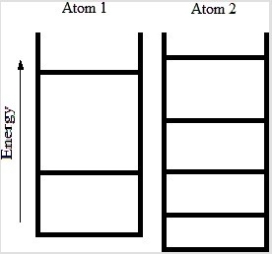 Based on the diagrams, which atom has the larger standard molar entropy at T = 298 K?
Based on the diagrams, which atom has the larger standard molar entropy at T = 298 K?
A) Atom 1 has the larger standard molar entropy because the energy of its ground state is larger than the energy of the ground state of atom 2.
B) Atom 2 has the larger standard molar entropy because the energy of its ground state is smaller than the energy of the ground state of atom 1.
C) Atom 1 has the larger standard molar entropy because it has fewer energy levels at lower energy values than atom 2.
D) Atom 2 has the larger standard molar entropy because it has more energy levels at lower energy values than atom 1.
E) Both atoms have the same value of the standard molar entropy at T = 298 K.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is a statement of the second law of thermodynamics?
A) The energy of the universe is constant.
B) The entropy of the universe is constant.
C) The enthalpy of the universe is constant.
D) The energy of the universe is decreasing.
E) The entropy of the universe is increasing.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Nitric oxide reacts with chlorine to form NOCl. 2NO(g) + Cl2(g) → 2NOCl(g) 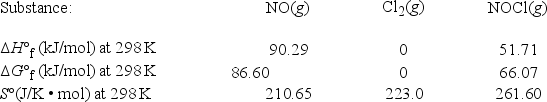 What is ΔG°rxn for this reaction at 550. K?
What is ΔG°rxn for this reaction at 550. K?
A) -143.76 kJ/mol
B) -78.78 kJ/mol
C) -22.24 kJ/mol
D) -10.6 kJ/mol
E) 66600 kJ/mol
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which reaction has the largest ΔS°?
A) 2N2H4(g) → 2NH3(g) + H2(g) + N2(g)
B) O2(g) + 2H2(g) → 2H2O(l)
C) O2(g) + 2H2(g) → 2H2O(g)
D) N2(g) + 3H2(g) → 2NH3(g)
E) 2NO(g) → N2O2(g)
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following process, which is true? 
A) ΔH > 0, ΔS > 0
B) ΔH > 0, ΔS < 0
C) ΔH < 0, ΔS > 0
D) ΔH < 0, ΔS < 0
E) ΔH > 0, ΔS = 0
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If a certain process has ΔSuniv > 0 at 25°C, the process may be described as
A) exothermic.
B) endothermic.
C) spontaneous.
D) moving rapidly toward equilibrium.
E) violating the second law of thermodynamics.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is a statement of the third law of thermodynamics?
A) The entropy of the universe is increasing.
B) The entropy of the universe is constant.
C) For a perfect crystalline solid, the entropy at T = 0 K is zero.
D) The energy of the universe is constant.
E) The entropy of all elements are zero.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
ΔSuniverse > 0 is the general condition for a ________ process.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A spontaneous endothermic reaction always
A) causes the surroundings to get colder.
B) bursts into flame.
C) requires a spark to initiate it.
D) releases heat to the surroundings.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔS° at 298 K for the following reaction? Fe2O3(s) + 3CO(g) → 3CO2(g) + 2Fe(s) 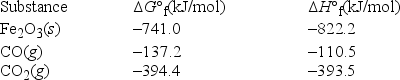
A) 12.7 J/K • mol
B) -12.7 J/K • mol
C) 527.9 J/K • mol
D) 157.2 J/K • mol
E) 4.6 J/K • mol
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A negative sign for ΔG indicates that, at constant T and P,
A) the reaction is exothermic.
B) the reaction is endothermic.
C) the reaction is fast.
D) the reaction is spontaneous.
E) ΔS of the system must be greater than zero.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Predict the sign of ΔS for the reaction O2(g) → 2O(g).
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which has the largest entropy?
A) C (diamond)
B) C (graphite)
C) H2O(l)
D) F2(l)
E) O2(g)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement is correct?
A) The standard entropy of a gas phase substance is less than the standard entropy of the same substance in the solid phase.
B) For two different substances in the same phase, the substance with the more complex structure has the lower entropy.
C) The more highly ordered allotrope of an element has a higher entropy.
D) The entropies of elements and compounds are always positive.
E) For a monatomic gas, the gas with the lower molar mass has the greater entropy.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
For a given reaction, a change in the temperature may result in a change in the sign of ΔG.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the thermodynamic condition for a spontaneous process at constant T and P?
A) ΔS > 0
B) ΔS < 0
C) ΔG < 0
D) ΔG > 0
E) ΔG° = 0
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
All elements in their standard state have standard entropies of formation equal to zero.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many types of rotations does a bent triatomic molecule exhibit?
A) 0
B) 1
C) 2
D) 3
E) 4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 107
Related Exams