A) -51.90 kJ/mol
B) -527.5 kJ/mol
C) -974.0 kJ/mol
D) -1502 kJ/mol
E) -2476 kJ/mol
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The heat of solution of ammonium nitrate is 26.2 kJ/mol. If a 5.368 g sample of NH4NO3 is added to 40.0 mL of water in a calorimeter at 23.5°C, what is the final temperature of the solution? The specific heat of water is 4.18 J/g·°C and the heat capacity of the calorimeter is 0.650 kJ/°C.
A) 14.3°C
B) 20.8°C
C) -7.7°C
D) 25.6°C
E) 21.4°C
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
The enthalpy of combustion of acetylene (C2H2) is described by C2H2(g) + 5/2O2(g) → 2CO2(g) + H2O(l) ΔH°rxn= -1299 kJ/mol Calculate the enthalpy of formation of acetylene, given the following enthalpies of formation: ΔH°f(CO2(g)) = -393.5 kJ/mol; ΔH°f(H2O(l)) = -285.8 kJ/mol
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When Karl Kaveman adds chilled grog to his new granite mug, he removes 10.9 kJ of energy from the mug. If it has a mass of 625 g and was at 25°C, what is its new temperature? Specific heat capacity of granite = 0.79 J/g·°C.
A) 3°C
B) 14°C
C) 22°C
D) 47°C
E) None of these choices is correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete the sentence: When heat is transferred to the system, the process is said to be ________, and the sign of q is ________.
A) exothermic, positive
B) exothermic, negative
C) endothermic, positive
D) endothermic, negative
E) forbidden, indeterminate
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The specific heat of gold is 0.129 J/g·°C. What is the molar heat capacity of gold?
A) 0.0394 J/mol·°C
B) 0.129 J/mol·°C
C) 25.4 J/mol·°C
D) 39.4 kJ/mol·°C
E) 197 J/mol·°C
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the change in internal energy in joules for a system that releases 154 J of heat and does 125 J of work on its surroundings?
A) -29 J
B) 279 J
C) 29 J
D) 1.23 J
E) -279 J
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A system which does work on the surroundings with no heat change has
A) w < 0, ΔU = 0.
B) w > 0, ΔU > 0.
C) w > 0, ΔU < 0.
D) w < 0, ΔU > 0.
E) w < 0, ΔU < 0.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Essay
a. Explain fully what is meant by the term "state function." b. (i) Give two examples of thermodynamic quantities which are state functions. (ii) Give two examples of thermodynamic quantities which are not state functions.
Correct Answer

verified
a. A state function depends only on the initial and final states, not on its history or the path followed in a process.
b. (i) internal energy, enthalpy (and possibly T, P, etc.)
(ii) heat and work
Correct Answer
verified
Short Answer
The sign of q when heat is released by the system is ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Benzene is a starting material in the synthesis of nylon fibers and polystyrene (Styrofoam) . Its specific heat capacity is 1.74 J/g·°C. If 16.7 kJ of energy is absorbed by a 225-g sample of benzene at 20.0°C, what is its final temperature?
A) -22.7°C
B) 37.7°C
C) 42.7°C
D) 62.7°C
E) 80.1°C
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many calories are in 854.3 J? (1 cal = 4.184 J)
A) 4.897 × 10-3 cal
B) 204.2 cal
C) 1.171 ×10-3 cal
D) 0.2390 cal
E) 3574 cal
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
The enthalpy of vaporization of a compound is always positive.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using Hess's law, what is the standard enthalpy of formation, ΔH°f of manganese(II) oxide, MnO(s) ? 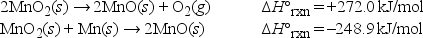
A) -520.9 kJ/mol
B) -396.5 kJ/mol
C) -384.9 kJ/mol
D) -147.6 kJ/mol
E) 24 kJ/mol
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many joules are in 1.20 ×103 calories? (1 cal = 4.184 J)
A) 8.33 × 10-4 J
B) 3.49 × 10-3 J
C) 2.39 × 10-1 J
D) 2.86 × 102 J
E) 5.02 × 103 J
G) C) and D)
Correct Answer

verified
E
Correct Answer
verified
Multiple Choice
Natural gas, or methane, is an important fuel. Combustion of one mole of methane releases 802.3 kJ of energy. How much energy does that represent in kcal? (1 cal = 4.184 J)
A) 1.92 × 10-1 kcal
B) 1.92 × 102 kcal
C) 3.36 × 103 kcal
D) 1.92 × 105 kcal
E) 3.36 × 106 kcal
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
All elements in their standard state have an enthalpy of formation equal to zero.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Suppose a 50-g block of silver (specific heat = 0.2350 J/g·°C) at 100°C is placed in contact with a 50-g block of iron (specific heat = 0.4494 J/g·°C) at 0°C, and the two blocks are insulated from the rest of the universe. The final temperature of the two blocks
A) will be higher than 50°C.
B) will be lower than 50°C.
C) will be exactly 50°C.
D) is unrelated to the composition of the blocks.
E) cannot be predicted.
G) A) and C)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
A common laboratory reaction is the neutralization of an acid with a base. When 50.0 mL of 0.500 M HCl at 25.0°C is added to 50.0 mL of 0.500 M NaOH at 25.0°C in a coffee cup calorimeter, the temperature of the mixture rises to 28.2°C. What is the heat of reaction per mole of acid? Assume the mixture has a specific heat capacity of 4.18 J/g·°C and that the densities of the reactant solutions are both 1.00 g/mL.
A) 670 J
B) 1300 J
C) 27 kJ
D) 54 kJ
E) > 100 kJ
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The dissolution of barium hydroxide in water is an exothermic process. Which statement is correct?
A) The enthalpy of solid barium hydroxide plus pure water is less than that of the solution, at the same temperature.
B) The enthalpy of solid barium hydroxide plus pure water is greater than that of the solution, at the same temperature.
C) The enthalpy of solid barium hydroxide plus pure water is the same as that of the solution, at the same temperature.
D) The temperature of the solution is lower than of the barium hydroxide and water before mixing.
E) When barium hydroxide dissolves in water, the system does work on the surroundings.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 141
Related Exams