Correct Answer

verified
Correct Answer
verified
Multiple Choice
A coastal climate is moderated primarily by which of the following properties of water? Water
A) is the universal solvent.
B) is cohesive and adhesive.
C) has a high heat of evaporation.
D) has a high surface tension.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
This system of chemicals, 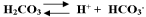 ,act as a buffer in the blood.If hydrogen ions are added to blood which of the following reactions would occur?
,act as a buffer in the blood.If hydrogen ions are added to blood which of the following reactions would occur?
A)
B)
D) undefined
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement is NOT true about ionic bonds?
A) One atom acts as an electron donor and another atom acts as an electron acceptor.
B) Electrons are completely lost or gained in ion formation.
C) An ion has the same number of electrons as a nonionic atom of the same element.
D) An ionic bond occurs between positive ions and negative ions.
E) A salt such as NaCl is formed by an ionic reaction.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The mass number refers to the number of ______ & ______ within an element.
A) protons & neutrons
B) protons & electrons
C) electrons & neutrons
D) protons & molecules
E) electrons & atoms
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Not Answered
Draw three water molecules and the hydrogen bonding that may occur between the molecules.Define hydrogen bonding and explain how and why it occurs.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Both 18O and 16O are found in nature.However,16O is the most common.Therefore,
A) these are different elements.
B) oxygen atoms can have eight or 10 neutrons.
C) 18O has two additional electrons in its outer shell.
D) 18O is the form of oxygen that provides living cells with life.
E) only the common form of 16O can bond with hydrogen atoms to form H2O.
G) C) and D)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
An orbital is best described as
A) the electron shell closest to the nucleus.
B) the outermost electron shell of an atom.
C) the volume of space in which electrons are most often found.
D) the original energy level of electrons in photosynthesis.
F) All of the above
Correct Answer

verified
C
Correct Answer
verified
Not Answered
All living things are 70 - 90% water.Use this graph to explain what characteristics of water protect living organisms from rapid temperature changes. 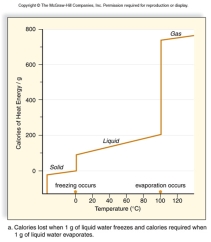
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is/are an atom,an isotope and an ion?
A) H+
B) 2H or deuterium
C) 3H or tritium
D) H2 or hydrogen gas
E) All of the choices are atoms,isotopes and ions.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
To determine the age of fairly recent fossils and organic artifacts,it is possible to analyze the amounts of the isotopes 14C and 14N,because over time the 14C-which originated in the atmosphere-breaks down into 14N.What net change occurred for this to happen?
A) The 14C lost an electron.
B) The 14C gained an electron.
C) The 14C lost a proton.
D) The 14C gained a proton.
E) The 14C gained a neutron.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Figure: -From the above table,it is apparent that:
A) triple bonds are stronger than double bonds; double bonds are stronger than single bonds.
B) triple bonds are weaker than double bonds; double bonds are weaker than single bonds.
C) carbon bonds are stronger than other bonds; hydrogen bonds are always weakest.
D) carbon forms only single bonds.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An abandoned Indiana coal mine spoil bank contains chunks of pyrite minerals.Under constant erosion and weathering,the pyrites leech large amounts of sulfuric acid (H2SO4) .The spoil banks are also mixed with large quantities of basic limestone and clay carbonates.What should occur over time?
A) The pH level will drop until all acid has washed out.
B) The pH level will remain at 7.0 because of constant washing with rain.
C) The pH level will remain at 7.0 because all acid will be immediately neutralized by bases.
D) The pH levels will be spotty and vary over time,first more acidic but drifting back toward 7.0.
E) Bases always dominate over acids.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An atom's atomic mass is best described as the mass of
A) the protons it contains.
B) the neutrons it contains.
C) electrons in the outermost shell.
D) protons and neutrons it contains.
E) protons and electrons it contains.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
All of the following are examples of damage caused by acid deposition from rain EXCEPT
A) leaching of aluminum from the soil into lakes which results in the formation of toxic methyl mercury from mercury in the lake sediments
B) weakens trees in the forests and kills seedlings
C) increased agricultural yields
D) damage to marble and limestone monuments
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution with a pH of 6 has ________ times _________ OH- than a solution with a pH of 10.
A) 40; more
B) 4000; less
C) 104; less
D) 4; less
E) 10-4 more
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution with a pH of 7.0 has _______ times ________ H+ than a solution of pH 10.
A) 30; more
B) 300; less
C) 103; more
D) 10-3; less
E) none of these are correct.
G) A) and E)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Figure:  -From the above table of radioisotopes and their properties,it is obvious that
-From the above table of radioisotopes and their properties,it is obvious that
A) the longer the half-life,the more energy emitted by the particles.
B) the longer the half-life,the less energy emitted by the particles.
C) radioisotopes of the same element must emit the same amount of energy in their emissions and decay at the same rate.
D) adjusted for time,radioisotopes emit the same amount of energy in their emissions.
E) energy and half-life are not directly related.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which type of covalent bond is the strongest?
A) single
B) double
C) triple
D) quadruple
E) all covalent bonds are equal in strength
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The blood buffer reactions described by H2CO3
 H+ + HCO3- indicates that
H+ + HCO3- indicates that
A) scientists are uncertain which direction the equation flows.
B) the reaction can flow either direction depending on whether there is an excess of hydrogen or hydroxide ions.
C) any reaction in one direction causes an immediate reverse reaction.
D) chemicals can swing wildly from acid to basic.
E) there is really no difference in chemistry whether a molecule is formed or dissociated.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 57
Related Exams