A) 6.46 × 10-25 J
B) 6.46 × 10-16 J
C) 2.46 × 10-4 J
D) 12.4 kJ
E) 246 kJ
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which element has the following ground-state electron configuration? [Ar]4s23d104p5
A) aresnic
B) bromine
C) iodine
D) selenium
E) tellerium
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the total number of electrons possible in the 6s orbital?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The orbital diagram for a ground-state nitrogen atom is 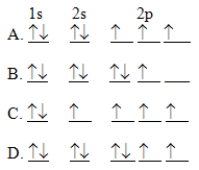
A) A
B) B
C) C
D) D
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The ground-state electron configuration of Cr, Mo, and Ag are exceptions to the Aufbau principle.Which of the following is the electron configuration for Mo?
A) [Kr]5s14d5
B) [Kr]5s24d4
C) [Xe]6s25d4
D) [Ar]4s24d4
E) [Kr]5s24d6
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
An FM radio station broadcasts at a frequency of 101.7 MHz.Calculate the wavelength of the broadcast signal in meters.(c = 2.9979 × 108 m/s)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The orbital diagram for a ground state carbon atom is 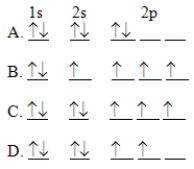
A) A
B) B
C) C
D) D
F) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The quantum numbers, n = 4, l = 1, ml = 1, ms = +1/2, represent an electron is a 4p subshell.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
A neon atom in its ground state will be diamagnetic.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
A photovoltaic cell converts light into electrical energy.Suppose a certain photovoltaic cell is only 63.5% efficient, in other words, that 63.5% of the light energy is ultimately recovered.If the energy output of this cell is used to heat water, how many 520 nm photons must be absorbed by the photovoltaic cell in order to heat 10.0 g of water from 20.0°C to 30.0°?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
"No two electrons in an atom can have the same four quantum numbers" is a statement of
A) the Pauli exclusion principle.
B) Bohr's equation.
C) Hund's rule.
D) de Broglie's relation.
E) Dalton's atomic theory.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which ground-state atom has an electron configuration described by the following orbital diagram? 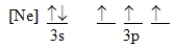
A) phosphorus
B) nitrogen
C) arsenic
D) vanadium
E) none of these
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which element has the following ground-state electron configuration? [Xe]6s24f145d4
A) W
B) Pm
C) Mo
D) Np
E) Re
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the maximum number of electrons in an atom that can have the following set of quantum numbers? n = 3 l = 2 ml = -2
A) 18
B) 10
C) 5
D) 2
E) 1
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
The colors of the visible spectrum are blue, green, orange, red, violet, and yellow.Of these colors, _______ has the longest wavelength.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many orbitals are allowed in a subshell if l = 2?
A) 1
B) 3
C) 5
D) 7
E) 9
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 121 - 136 of 136
Related Exams