A) 43.4 kJ/mol
B) 52.5 kJ/mol
C) 70.6 kJ/mol
D) 1.00 102 kJ/mol
E) None of these choices is correct.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following has a boiling point which does not fit the general trend?
A) NH3
B) PH3
C) AsH3
D) SbH3
E) BiH3
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
In the packing of identical atoms with cubic unit cells,the packing efficiency increases as the coordination number increases.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following substances will have hydrogen bonds between molecules?
A) (CH3) 3N
B) CH3-O-CH3
C) CH3CH2-OH
D) CH3CH2-F
E) HI
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which of the following compounds will the molecules not form hydrogen bonds with each other? 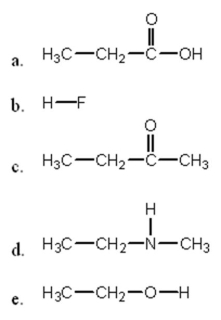
A) a
B) b
C) c
D) d
E) e
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pairs of substances is arranged so that the one with higher viscosity is listed first? 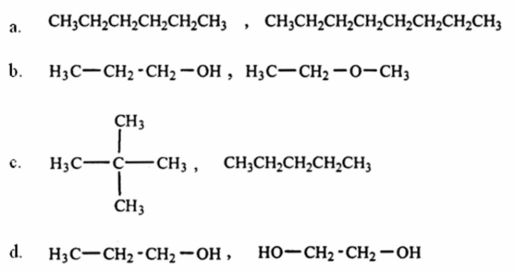
A) a
B) b
C) c
D) d
E) None of these choices is correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In an ionic solid MX consisting of the monatomic ions,M+ and X-,the coordination number of M+ is _______________.
A) 1
B) 2
C) 6
D) 8
E) impossible to predict without knowing the crystal structure of MX
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Diethyl ether,used as a solvent for extraction of organic compounds from aqueous solutions,has a high vapor pressure which makes it a potential fire hazard in laboratories in which it is used.How much energy is released when 100.0 g is cooled from 53.0°C to 10.0°C?
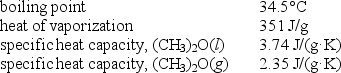
A) 10.1 kJ
B) 13.1 kJ
C) 16.1 kJ
D) 45.2 kJ
E) 48.6 kJ
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following atoms should have the smallest polarizability?
A) Si
B) S
C) Te
D) Bi
E) Br
G) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 81 - 89 of 89
Related Exams