A) 2.0 days
B) 16 days
C) 32 days
D) 128 days
E) None of these choices is correct.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The isotope 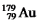 Has a half-life of 7.5 seconds.If a sample contains 144 atoms of
Has a half-life of 7.5 seconds.If a sample contains 144 atoms of 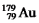 ,approximately how many such atoms were there present 30 seconds earlier?
,approximately how many such atoms were there present 30 seconds earlier?
A) 576
B) 1152
C) 2304
D) 4320
E) 4.30 108
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A certain isotope has a specific activity of 7.29 10-4 Ci/g.How many particles will a 75.0 mg sample emit in one hour?
A) 9.99 104
B) 2.02 106
C) 7.28 109
D) 1.29 1012
E) None of these choices is correct.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
After 4 half-lives,the fraction of a radioactive isotope which still remains is approximately one eighth.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the missing species in the following nuclear transmutation. 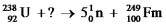
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Radioactive decay follows zero-order kinetics.
B) False
Correct Answer

verified
Correct Answer
verified
Essay
Fill in missing sub- and superscripts for all particles to complete the following equation for positron decay. 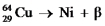
Correct Answer

verified
Correct Answer
verified
Essay
Briefly explain the relationship between the rad and the rem as units of radiation dosage.
Correct Answer

verified
The rad (radiation absorbed dose)measure...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
Write a complete,balanced equation to represent the electron capture decay of argon-37.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the nuclide that completes the following nuclear reaction. 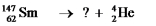
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A pure sample of tritium,3H,was prepared and sealed in a container for a number of years.Tritium undergoes decay with a half-life of 12.32 years.How long has the container been sealed if analysis of the contents shows there are 5.25 mol of 3H and 6.35 mol of 3He present?
A) 2.34 y
B) 3.38 y
C) 9.77 y
D) 14.1 y
E) 25.6 y
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following equations correctly represents electron capture by the 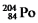 Nucleus?
Nucleus?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following materials is put into a nuclear reactor to slow the chain reaction?
A) heavy water
B) moderators
C) control rods
D) reflectors
E) chlorine
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The isotope 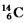 Is unstable.This is predictable because
Is unstable.This is predictable because
A) N/Z 1.
B) N/Z is relatively low and Z < 20.
C) N/Z is relatively large and Z < 20.
D) Z is small.
E) N is large.
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
The binding energy per nucleon reaches a maximum for the isotope 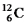 .
.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An isotope with a low value of N/Z will generally decay through
A) ( decay.)
B) 9 decay.)
C) ( decay.)
D) electron capture.
E) spontaneous fission.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Fill in missing sub- and superscripts for all particles to complete the following equation for beta decay. 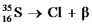
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the nuclide that completes the following nuclear reaction. 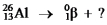
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following equations correctly represents alpha decay of 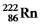 ?
?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
Write a complete,balanced equation to represent the formation of manganese-55 by the beta decay of another nuclide.
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 75
Related Exams