B) False
Correct Answer

verified
Correct Answer
verified
Essay
Draw a fully labeled phase diagram (P versus T) of a substance whose solid phase can melt due to applied pressure (i.e., solid is less dense than liquid). Clearly label the triple point and the critical temperature on your diagram.
Correct Answer

verified
Correct Answer
verified
True/False
Only molecules which do not have dipole moments can experience dispersion forces.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
Iron has a body-centered cubic unit cell, and a density of 7.87 g/cm3. Calculate the edge length of the unit cell, in pm. (The atomic mass of iron is 55.85 amu. Also, 1 amu = 1.661 × 10-24 g.)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Lead crystallizes in the face-centered cubic lattice. What is the coordination number for Pb?
A) 4
B) 6
C) 8
D) 10
E) 12
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Octane has a vapor pressure of 40. torr at 45.1°C and 400. torr at 104.0°C. What is its heat of vaporization?
A) 39.0 kJ/mol
B) 46.0 kJ/mol
C) 590 kJ/mol
D) 710 kJ/mol
E) none of the above
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following terms refers to the resistance of a liquid to flow?
A) surface tension
B) capillary action
C) viscosity
D) adhesion
E) cohesion
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The surface tension of water is lowered when a detergent is present in solution.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
Liquid ammonia boils at -33.4°C and has a heat of vaporization of 23.5 kJ/mol. Calculate its vapor pressure at -50.0°C.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A temperature increase causes __________________ in the conductivity of a semiconductor.
A) a decrease
B) an increase
C) a modulation
D) an increase or decrease (depending on the semiconductor)
E) no change
G) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
The density of solid sodium chloride, NaCl, is 2.17 g/cm3. Use your knowledge of the sodium chloride lattice to calculate the spacing between Na+ and Cl- nearest neighbors, in cm. (Atomic masses (amu) are: Na, 22.99; Cl, 35.45. Also, 1 amu = 1.661 × 10-24 g)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The energy gap between the conduction band and the valence band is large for
A) conductors.
B) semiconductors.
C) superconductors.
D) insulators.
E) alloys.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following phase diagram and identify the process occurring as one goes from point C to point D. 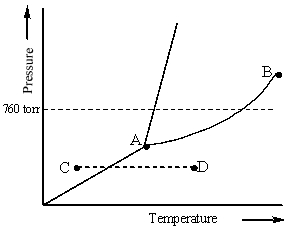
A) increasing temperature with a phase change from solid to liquid
B) increasing temperature with a phase change from solid to vapor
C) increasing temperature with a phase change from liquid to vapor
D) increasing temperature with no phase change
E) increasing temperature beyond the critical point
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Essay
How do the electrical properties of semiconductors differ from those of metals?
Correct Answer

verified
Metals are good conductors who...View Answer
Show Answer
Correct Answer
verified
View Answer
True/False
The maximum number of phases of a single substance which can coexist in equilibrium is two.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following properties measures the energy needed to increase the surface area of a liquid?
A) capillary action
B) surface tension
C) viscosity
D) cohesion
E) specific elasticity
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the pair of substances in which the one with the lower vapor pressure at a given temperature is listed first.
A) ![]() ,
, ![]()
B) ![]() ,
, ![]()
C) ![]() ,
, ![]()
D) ![]() ,
, ![]()
E) ![]() ,
, ![]()
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following liquid substances would you expect to have the lowest surface tension?
A) Pb
B) CH3OCH3
C) HOCH2CH2OH
D) H2O
E) CH3CH2OH
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Polonium crystallizes in the simple cubic lattice. What is the coordination number for Po?
A) 3
B) 4
C) 6
D) 8
E) 12
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following atoms should have the greatest polarizability?
A) F
B) Br
C) Po
D) Pb
E) He
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 111
Related Exams