A) < 4.00
B) 4.63
C) 4.87
D) 5.02
E) > 5.50
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A lab technician adds 0.015 mol of KOH to 1.00 L of 0.0010 M Ca(NO3) 2. Ksp = 6.5 × 10-6 for Ca(OH) 2) . Which of the following statements is correct?
A) Calcium hydroxide precipitates until the solution is saturated.
B) The solution is unsaturated and no precipitate forms.
C) The concentration of calcium ions is reduced by the addition of the hydroxide ions.
D) One must know Ksp for calcium nitrate to make meaningful predictions on this system.
E) The presence of KOH will raise the solubility of Ca(NO3) 2.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following substances has the greatest solubility in water?
A) MgCO3, Ksp = 3.5 × 10-8
B) NiCO3, Ksp = 1.3 × 10-7
C) AgIO3, Ksp = 3.1 × 10-8
D) CuBr, Ksp = 5.0 × 10-9
E) AgCN, Ksp = 2.2 × 10-16
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following indicators would be the best to use when 0.050 M benzoic acid (Ka = 6.6 × 10-5) is titrated with 0.05 M NaOH?
A) bromphenol blue, pH range: 3.0-4.5
B) bromcresol green, pH range: 3.8-5.4
C) alizarin, pH range: 5.7-7.2
D) phenol red, pH range: 6.9-8.2
E) phenolphthalein, pH range: 8.0-10.1
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 25.0-mL sample of 0.35 M HCOOH is titrated with 0.20 M KOH. What is the pH of the solution after 25.0 mL of KOH has been added to the acid? Ka = 1.77 × 10-4
A) 4.00
B) 3.88
C) 3.63
D) 3.51
E) 3.47
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The solubility of silver chloride _______________ when dilute nitric is added to it.
A) increases
B) decreases
C) does not change
D) first increases, then decreases
E) first decreases, then increases
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution is prepared by adding 4.50 mol of sodium hydroxide to 1.00 L of 1.00 M Co(NO3) 2. What is the equilibrium concentration of cobalt ions? Kf = 5.0 × 109 for Co(OH) 42-
A) 1.1 × 10-2 M
B) 1.4 × 10-5 M
C) 3.2 × 10-9 M
D) 2.0 × 10-10 M
E) 4.9 × 10-13 M
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the pH of 375 mL of solution containing 0.150 mol of propenoic acid (HA) and 0.250 mol of sodium propenoate (NaA)? (Ka for propenoic acid is 5.52 × 10-5.)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 20.0-mL sample of 0.30 M HClO was titrated with 0.30 M NaOH. The following data were collected during the titration.  What is the Ka for HClO?
What is the Ka for HClO?
A) 1.1 × 10-7
B) 3.5 × 10-8
C) 1.2 × 10-8
D) 4.9 × 10-11
E) none of the above
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A buffer is prepared by adding 100 mL of 0.50 M sodium hydroxide to 100 mL of 0.75 M propanoic acid. Is this a buffer solution, and if so, what is its pH?
A) It is a buffer, pH > pKa of propanoic acid.
B) It is a buffer, pH < pKa of propanoic acid.
C) It is a buffer, pH = pKa of propanoic acid.
D) It is a buffer, pH = pKb of sodium propanoate.
E) Since sodium hydroxide is a strong base, this is not a buffer.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Essay
Use a carefully drawn and labeled diagram of the titration curve to illustrate the titration of a weak diprotic acid, in which Ka1 and Ka2 are substantially different, with a strong base (base is the titrant). Label as many features of the diagram as possible.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 20.0-mL sample of 0.25 M HNO3 is titrated with 0.15 M NaOH. What is the pH of the solution after 30.0 mL of NaOH have been added to the acid?
A) 2.00
B) 1.60
C) 1.05
D) 1.00
E) none of the above
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the dissolution of MnS in water
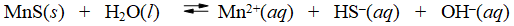 How is the solubility of manganese(II) sulfide affected by the addition of aqueous potassium hydroxide to the system?
How is the solubility of manganese(II) sulfide affected by the addition of aqueous potassium hydroxide to the system?
A) The solubility will be unchanged.
B) The solubility will decrease.
C) The solubility will increase.
D) The amount of KOH added must be known before its effect can be predicted.
E) The pKa of H2S is needed before a reliable prediction can be made.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When a strong acid is titrated with a weak base, the pH at the equivalence point
A) is greater than 7.0.
B) is equal to 7.0.
C) is less than 7.0.
D) is equal to the pKa of the acid.
E) is equal to the pKb of the base.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following aqueous mixtures would be a buffer system?
A) HCl, NaCl
B) HNO3, NaNO3
C) H3PO4, H2PO4-
D) H2SO4, CH3COOH
E) NH3, NaOH
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Assuming that the total volume does not change after 0.200 g of KCl is added to 1.0 L of a saturated aqueous solution of AgCl, calculate the number of moles of Ag+ ion in the solution after equilibrium has been reestablished. For AgCl, Ksp = 1.8 × 10- 10.
A) 1.8 × 10-10 mol Ag+
B) 9.0 × 10-10 mol Ag+
C) 9.0 × 10-9 mol Ag+
D) 6.7 × 10-8 mol Ag+
E) 1.3 × 10-5 mol Ag+
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the [H3O+] in a buffer that consists of 0.30 M HCOOH and 0.20 M HCOONa? For HCOOH, Ka = 1.7 × 10-4
A) 1.1 × 10-4 M
B) 2.6 × 10-4 M
C) 4.3 × 10-4 M
D) 6.7 × 10-5 M
E) none of the above
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following information to calculate the solubility product constant, Ksp, for PbCl2. A saturated solution of PbCl2 in water was prepared and filtered. From the filtrate, 1.0 L was measured out into a beaker and evaporated to dryness. The solid PbCl2 residue recovered in the beaker amounted to 0.0162 moles.
A) Ksp = 6.9 × 10-8
B) Ksp = 4.3 × 10-6
C) Ksp = 1.7 × 10-5
D) Ksp = 2.6 × 10-4
E) Ksp = 3.2 × 10-2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the [H3O+] in a solution that consists of 1.5 M NH3 and 2.5 NH4Cl? Kb = 1.8 × 10-5
A) 1.1 × 10-5 M
B) 3.0 × 10-6 M
C) 3.3 × 10-9 M
D) 9.3 × 10-10 M
E) none of the above
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The salts X(NO3) 2 and Y(NO3) 2 (where X+ and Y+ are metal ions) are dissolved in water to give a solution which is 0.1 M in each of them. Which of the answers gives the concentration of chloride ions will precipitate the most YCl2 without precipitating any XCl2? Given Ksp values: XCl2, 2 × 10-5 YCl2, 1 × 10-10
A) 1 M Cl-
B) 0.1 M Cl-
C) 0.01 M Cl-
D) 0.001 M Cl-
E) 0.0001 M Cl-
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 119
Related Exams