A) 1
B) 2
C) 4
D) 6
E) 8
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following species could exist as isomers?
A) [Co(H2O) 4Cl2]+
B) [Pt(NH3) Br3]-
C) [Pt(en) Cl2]
D) [Pt(NH3) 3Cl]+
E) none of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ligands could participate in linkage isomerism?
A) NH3
B) H2O
C) NH4+
D) NO2-
E) ethylenediamine
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following octahedral complex structures, each involving ethylene diamine and two different, unidentate ligands X and Y. 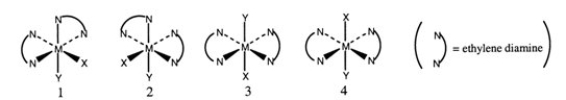 Which one, if any, of the following is a pair of optical isomers?
Which one, if any, of the following is a pair of optical isomers?
A) 1 and 2
B) 1 and 3
C) 1 and 4
D) 3 and 4
E) none of the above
G) A) and B)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
A feature of transition metal chemistry is that these elements exhibit multiple oxidation states. Which one of the following elements exhibits the smallest number of different oxidation states?
A) Ti
B) Cr
C) Mn
D) Co
E) Zn
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The crystal field splitting energy, ,
A) is larger for tetrahedral complexes than for octahedral complexes.
B) depends on the metal but not on the ligand.
C) determines the color of a complex.
D) is larger for ionic ligands like chloride than for molecular ligands like carbon monoxide, CO.
E) determines the charge of a complex.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the oxidation states of chromium has the largest valence-state electronegativity?
A) chromium(0)
B) chromium(II)
C) chromium(III)
D) chromium(IV)
E) chromium(VI)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A certain transition metal complex has the formula MX42+. If the metal ion has a d8 electron configuration, what is the shape of the complex?
A) octahedral
B) square pyramid
C) tetrahedral
D) trigonal pyramid
E) square planar
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the highest possible oxidation state for palladium, Pd?
A) +1
B) +2
C) +3
D) +4
E) +6
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many unpaired electrons will there be in a high-spin octahedral complex of Fe(II) ?
A) 0
B) 2
C) 4
D) 6
E) none of the above
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the presence of a strong octahedral ligand field, the number of unpaired electrons in Co(III) will be
A) 0.
B) 2.
C) 4.
D) 6.
E) none of the above.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following octahedral complex structures, each involving ethylene diamine and two different, unidentate ligands X and Y. 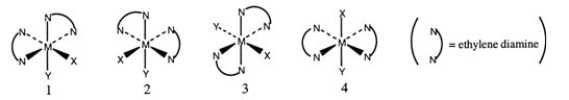 Which one of the following statements about these structures is incorrect?
Which one of the following statements about these structures is incorrect?
A) Structures 1 and 2 are optical isomers.
B) Structures 1 and 3 are optical isomers.
C) Structures 1 and 3 are different complexes.
D) Structures 1 and 4 are geometrical isomers.
E) Structures 3 and 4 are the same complex.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the ethylenediaminetetraacetate ion (EDTA4-) forms a complex with a transition metal ion, how many electrons does it normally donate to the metal?
A) 4
B) 6
C) 8
D) 10
E) 12
G) None of the above
Correct Answer

verified
Correct Answer
verified
Essay
a. How many unpaired 3d electrons will there be in (i) high and (ii) low-spin complexes of Co(II)? b. How can high and low-spin complexes be recognized and distinguished experimentally?
Correct Answer

verified
a. (i) high-spin, 3 unpaired electrons
(ii) low-spin, 1 unpaired electron
b. Unpaired electrons interact strongly with a magnetic field, and this property is used to determine the number of such electrons in a complex.
Correct Answer
verified
Multiple Choice
Chromium and manganese are among the transition elements that form several different oxides. Which of the following statements characterize these oxides?
A) As the oxidation number on the metal increases, the valence-state electronegativity increases and the oxides change from acidic to basic.
B) As the oxidation number on the metal increases, the valence-state electronegativity increases and the oxides change from basic to acidic.
C) As the oxidation number on the metal increases, the valence-state electronegativity decreases and the oxides change from acidic to basic.
D) As the oxidation number on the metal increases, the valence-state electronegativity decreases and the oxides change from basic to acidic.
E) None of the above statements is correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The ground state electronic configuration of Zn2+ is
A) [Ar]4s23d8.
B) [Ar]4s23d10.
C) [Ar]4s13d9.
D) [Ar]3d10.
E) [Ar]3d8.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the systematic name for [CoCl3(H2O) ]-.
A) cobalt(II) chloride monohydrate
B) aquatrichlorocobalt(II)
C) aquatrichlorocobaltate(II)
D) aquatrichlorocobaltite(I)
E) none of the above
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ions is least likely to form colored compounds?
A) Mn2+
B) Cr5+
C) Sc3+
D) Fe3+
E) Co2+
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following normally acts as a bidentate ligand in complexes with transition metal ions?
A) CN-
B) EDTA4-
C) SCN-
D) ethylene diamine
E) ethylene, C2H4
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Essay
a. Explain how the crystal field theory can use the magnitude of the splitting energy to provide an explanation of the color and magnetic properties of octahedral complexes. b. In promoting an electron from the t2g set of orbitals to the eg set, an octahedral complex absorbs a photon with a wavelength of 523 nm. Calculate the value of in the complex, in kJ/mol.
Correct Answer

verified
a. The energy needed for a photon to excite an electron from the lower to the higher set of the split d orbitals, will increase as F1F1F1S1F1F1F10 increases. This will mean the complex absorbs at shorter wavelengths, influencing its color. Also, a large splitting energy F1F1F1S1F1F1F10 will tend to produce low-spin complexes.
b. 229 kJ/mol
Correct Answer
verified
Showing 1 - 20 of 92
Related Exams