A) CO2
B) HCl
C) H2O
D) NH3
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is not an acceptable Lewis structure for the anion CH2NCO-? 
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When forming molecular orbitals from atomic orbitals, what is the order of increasing C-H bond strength for the following. 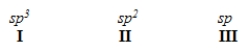
A) II < I < III
B) III < I < II
C) III < II < I
D) I < II < III
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about resonance structures is true?
A) Resonance structures have the same placement of electrons but different arrangement of atoms.
B) Resonance structures have the same placement of atoms but different arrangement of electrons.
C) Resonance structures have the same placement of atoms and the same arrangement of electrons.
D) Resonance structures have different placement of atoms and different arrangement of electrons.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the appropriate conversion of (CH3) 4C to a skeletal structure? 
A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which structure is the hybridization incorrect? 
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many constitutional isomers are there for a molecule having the molecular formula C3H6?
A) 1
B) 2
C) 3
D) 4
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What 2 things will change between two resonance structures?
A) The position of multiple bonds and non-bonded electrons.
B) The position of multiple bonds and single bonds.
C) The placement of atoms and single bonds.
D) The placement of atoms and non-bonded electrons.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which atomic orbitals overlap to form the carbon-carbon and bonding molecular orbitals of ethylene, H2C=CH2?
A) Csp3 + Csp3, and C2p + C2p
B) Csp3 + Csp3, and Csp2 + Csp2
C) Csp2 + Csp2, and C2p + C2p
D) Csp2 + Csp2, and Csp2 + Csp2
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules has non-polar covalent bonds?
A) CO2
B) N2
C) CCl4
D) HF
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the correct Lewis structure for acetic acid (CH3CO2H) ? 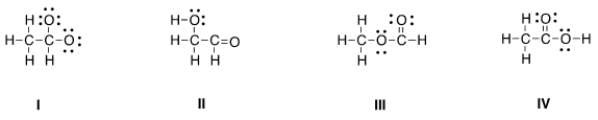
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements correctly describes the typical number of bonds for carbon, nitrogen, and oxygen in most neutral organic molecules?
A) Carbon forms 4 covalent bonds, nitrogen forms 2 covalent bonds and oxygen forms 3 covalent bonds.
B) Carbon forms 4 covalent bonds, nitrogen forms 3 covalent bonds and oxygen forms 2 covalent bonds.
C) Carbon forms 4 covalent bonds, nitrogen forms 5 covalent bonds and oxygen forms 2 covalent bonds.
D) Carbon forms 4 covalent bonds, nitrogen forms 5 covalent bonds and oxygen forms 4 covalent bonds.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the condensed formula of the compound below? 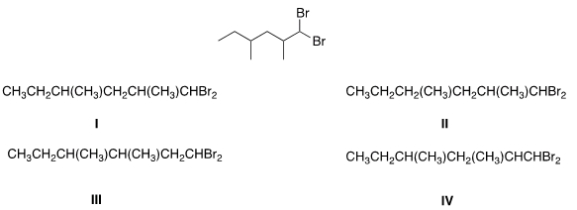
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following atoms in order of decreasing electronegativity, putting the most electronegative first.
A) I > IV > II > III
B) II > III > IV > I
C) III > IV > II > I
D) III > II > IV > I
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the approximate bond angle for the C-C-N bond in acetonitrile, CH3CN?
A) 90°
B) 109.5°
C) 120°
D) 180°
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the ground-state electronic configuration of a chlorine anion (Cl-) ?
A) 1s2, 2s2, 2p6
B) 1s2, 2s2, 2p6, 3s2, 3p6
C) 1s2, 2s2, 2p6, 3s2, 3p5
D) 1s2, 2s2, 2p6, 3s2, 3p4
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following atoms in order of increasing electronegativity, putting the least electronegative first.
A) I < II < III < IV
B) I < IV < II < III
C) III < II < IV < I
D) I < II < IV < III
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which atomic orbitals overlap to form the carbon-carbon bonding molecular orbital of acetylene, C2H2?
A) Csp2 + Csp2
B) Csp + Csp
C) Csp3 + Csp3
D) C2p + C2p
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about bonding is true?
A) Covalent bonds result from the transfer of electrons from one element to another.
B) Ionic bonds result from the transfer of electrons from a metal to a non-metal.
C) Ionic bonds result from the sharing of electrons between two non-metals.
D) Covalent bonds result from the sharing of electrons between two metals.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the approximate H-C-O bond angle in formaldehyde, H2CO?
A) 90°
B) 109.5°
C) 120°
D) 180°
F) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 70
Related Exams