A) an essential element in the human body.
B) toxic because it results in an increase in the production of ATP.
C) toxic because it binds to the sulfhydryl group in proteins and enzymes.
D) used in high concentrations in ancient cosmetics.
E) None of these choices is correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which,if any,of the following generalized formulas does not exist for interhalogen compounds? (X and Y represent different halogens.)
A) XY
B) XY2
C) XY3
D) XY5
E) All may represent stable interhalogen compounds.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What are the products of the reaction of strontium hydride and water? SrH2(s) + H2O(l) →
A) Sr2+(aq) + H2(g) + O2(g)
B) Sr2+(aq) + H2(g) + OH-(aq)
C) Sr(s) + H2(g) + OH-(aq)
D) Sr(s) + H3O+(aq)
E) Sr2+(aq) + H-(aq)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The chemical that ranks first in production among all industrial chemicals is
A) NH3,ammonia.
B) H3PO4,phosphoric acid.
C) NaOH,sodium hydroxide.
D) Na2CO3,sodium carbonate.
E) H2SO4,sulfuric acid.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
What has metal and nonmetal characteristics?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which best represents the allotrope(s) of oxygen?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
__________________ is the addition of molecular hydrogen to compounds containing multiple bonds (double or triple).
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below is a representation of a sample of gas with molecules having the formula XO2.Which element could NOT be represented by X? 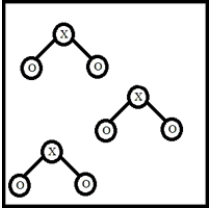
A) N
B) S
C) C
D) O
E) Cl
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In their elemental forms,the halogens are
A) strong oxidizing agents.
B) strong reducing agents.
C) strong acids.
D) strong bases.
E) amphoteric.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of these species exists as a diatomic element at room temperature?
A) Na
B) N
C) B
D) Ar
E) P
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a commercial application of phosphate compounds?
A) Detergents
B) Toothpaste
C) Fertilizers
D) Flame retardants
E) All of the answers are correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
Consider the oxides XO2,where X is a main group element.Identify X in each of the following cases: a.XO2 is a natural component of the atmosphere,implicated in global warming. b.XO2 is a toxic,brown gas and a component of photochemical smog. c.XO2 is a colorless,toxic gas,implicated in acid rain.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is not a product of the reaction of zinc metal with nitric acid?
A) NH4+
B) H2O
C) H2
D) Zn2+
E) All of the species are products.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which oxide is an amphoteric oxide?
A) Na2O
B) Al2O3
C) BaO
D) SiO2
E) More that one of these oxides are amphoteric.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these compounds does not follow the octet rule?
A) CO
B) CO2
C) NF3
D) NO
E) CF4
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What are the products of the reaction of potassium hydride and water? KH(s) + H2O(l) →
A) K(s) + H2(g) + OH-(aq)
B) K(s) + H2(g) + O2(g)
C) KOH(s) + O2(g)
D) K+(aq) + H2(g) + O2(g)
E) K+(aq) + H2(g) + OH-(aq)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many lone electron pairs are in the N2 molecule?
A) 1
B) 4
C) 3
D) 5
E) 2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrogen forms metallic (interstitial) hydrides with the d and f transition elements.Which of the following statements is correct?
A) These substances have distinct stoichiometric formulas like ionic hydrides.
B) Hydrogen forms bonds with the metals by donating its electron to the valence band of the metal.
C) Hydrogen molecules and atoms occupy holes within the crystal structure of the metal.
D) These substances are poor catalysts.
E) These hydrides are stabilized by hydrogen bonding forces.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these has a nonpolar covalent bond (i.e.,pure covalent) ?
A) Se-I
B) Na-Br
C) H-F
D) Cl-Cl
E) H-Cl
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Nonmetals are more electropositive than metals.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 119
Related Exams