A) CH3NH2,Kb = 4.4 × 10-4
B) C5H5N,Kb = 1.7 × 10-9
C) H2NCONH2,Kb = 1.5 × 10-14
D) NH3,Kb = 1.8 × 10-5
E) C2H5NH2,Kb = 5.6 × 10-4
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the concentration of H+ in a 0.025 M HCl solution?
A) 0
B) 0.013 M
C) 0.025 M
D) 0.050 M
E) 0.010 M
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the conjugate base of sulfuric acid?
A) HSO4-
B) H3O+
C) OH-
D) SO42-
E) H3SO4+
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Find the pH of a 0.183 M aqueous solution of hypobromous acid (HOBr) ,for which Ka = 2.06 ×10-9.
A) 4.712
B) 8.686
C) 0.738
D) 4.576
E) 9.288
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the conjugate acid of HS-?
A) HS2
B) S2-
C) HS
D) H2S
E) SOH
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Acid strength increases in the series HCN < HF < HSO4-.Which of these species is the strongest base?
A) H2SO4
B) SO42-
C) F-
D) CN -
E) HSO4-
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
If a strong acid such as HCl is diluted sufficiently with water,the pH will be higher than 7.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the conjugate acid of the hydroxide ion?
A) H3O+
B) OH-
C) H3O
D) OH
E) H2O
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the correct statement?
A) A polyprotic acid has more than two ionizable protons.
B) H2O is a diprotic acid.
C) All protons are lost from a polyprotic acid at the same time.
D) NH3 is a diprotic acid.
E) NH3 is a polyprotic acid.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Butyric acid is responsible for the odor in rancid butter.A solution of 0.25 M butyric acid has a pH of 2.71.What is the Ka for the acid?
A) 0.36
B) 2.4 × 10-2
C) 7.8 × 10-3
D) 1.5 × 10-5
E) 1.9 × 10-3
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
A hydrohalic acid is a binary acid containing a halogen.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution is prepared by adding 0.10 mol of potassium chloride,KCl,to 1.00 L of water.Which statement about the solution is correct?
A) The solution is basic.
B) The solution is neutral.
C) The solution is acidic.
D) The solution is a strong oxidant.
E) The values for Ka and Kb for the species in solution must be known before a prediction can be made.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction of a strong acid with water results in
A) a weak conjugate acid.
B) a strong conjugate base.
C) a weak conjugate base.
D) a strong base.
E) pure water.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of these salts will form a basic solution upon dissolving in water?
A) NaCl
B) NaNO2
C) NH4NO3
D) KBr
E) AlCl3
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the weakest acid?
A) HBrO4
B) HBr
C) HBrO2
D) HBrO
E) HBrO3
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ammonium chloride is used as an electrolyte in dry cells.Which statement about a 0.10 M solution of NH4Cl is correct?
A) The solution is weakly basic.
B) The solution is strongly basic.
C) The solution is neutral.
D) The solution is acidic.
E) The values for Ka and Kb for the species in solution must be known before a prediction can be made.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below is a representation of an aqueous solution of a weak acid HA at equilibrium.(Each circle represents 1.0 mmol of atoms,and the volume of the box is 1.0 L.Solvent water molecules are not shown for clarity.) 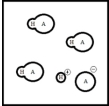 What is the percent ionization of HA?
What is the percent ionization of HA?
A) 100%
B) 50%
C) 25%
D) 12.5%
E) Cannot be calculated without knowledge of Ka
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH of a 0.050 M triethylamine,(C2H5) 3N,solution? Kb for triethylamine is 5.3 × 10-4.
A) 11.69
B) 8.68
C) 5.32
D) 2.31
E) 1.30
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the concentration of OH- in a 0.083 M NaOH solution?
A) 0.083 M
B) 1.21 × 10-13 M
C) 8.30 × 10-16 M
D) 7 M
E) 8.30 × 1012 M
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
RCOOH is an organic compound which is in the ______________ family.
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 133
Related Exams