A) There will be no effect.
B) More ammonia will be produced at the expense of hydrogen and nitrogen.
C) Hydrogen and nitrogen will be produced at the expense of ammonia.
D) The equilibrium constant will increase.
E) The equilibrium constant will decrease.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A quantity of liquid methanol,CH3OH,is introduced into a rigid 3.00-L vessel,the vessel is sealed,and the temperature is raised to 500 K.At this temperature,the methanol vaporizes and decomposes according to the reaction CH3OH(g)  CO(g) + 2H2(g) ,Kc = 6.90 ×10-2
If the concentration of H2 in the equilibrium mixture is 0.426 M,what mass of methanol was initially introduced into the vessel?
CO(g) + 2H2(g) ,Kc = 6.90 ×10-2
If the concentration of H2 in the equilibrium mixture is 0.426 M,what mass of methanol was initially introduced into the vessel?
A) 147 g
B) 74.3 g
C) 33.9 g
D) 49.0 g
E) 24.8 g
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Nitric oxide and bromine were allowed to react in a sealed container.When equilibrium was reached,the following partial pressures of three gases were measured: NO: 0.526 atm; Br2 :1.59 atm; NOBr: 7.68 atm.Calculate KP for the reaction. 2NO(g) + Br2(g)  2NOBr(g)
2NOBr(g)
A) 7.45 × 10-3
B) 0.109
C) 9.18
D) 91.8
E) 134
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The equilibrium constant,KP,has a value of 6.5 × 10-4 at 308 K for the reaction of nitrogen monoxide with chlorine. 2NO(g) + Cl2(g)  2NOCl(g)
What is the value of Kc? (R = 0.08206 L • atm/K • mol)
2NOCl(g)
What is the value of Kc? (R = 0.08206 L • atm/K • mol)
A) 2.5 × 10-7
B) 6.5 × 10-4
C) 1.6 × 10-2
D) 1.7
E) None of these choices is correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the endothermic reaction A2(g)  2A(g) ,a snapshot of an equilibrium mixture of A(g) and A2(g) at low temperature may look as follows.(Each circle represents 1.0 mol of A atoms,and the volume of the box is 1.0 L.)
2A(g) ,a snapshot of an equilibrium mixture of A(g) and A2(g) at low temperature may look as follows.(Each circle represents 1.0 mol of A atoms,and the volume of the box is 1.0 L.) 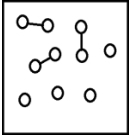 If the system pressure is lowered,what might the new equilibrium system look like?
If the system pressure is lowered,what might the new equilibrium system look like?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the correct equilibrium constant expression for the following reaction? 2BrCl3(g) ![Which is the correct equilibrium constant expression for the following reaction? 2BrCl<sub>3</sub>(g) Br<sub>2</sub>(g) + 3Cl<sub>2</sub>(g) A) K<sub>c</sub> = [Br<sub>2</sub>] [Cl<sub>2</sub>]/[BrCl<sub>3</sub>] B) K<sub>c</sub> = [Br<sub>2</sub>] [Cl<sub>2</sub>]<sup>5</sup>/[BrCl<sub>3</sub>]<sup>2</sup> C) K<sub>c</sub> = [Br<sub>2</sub>] [Cl<sub>2</sub>]<sup>3</sup>/[BrCl<sub>3</sub>]<sup>2</sup> D) K<sub>c</sub> = [BrCl<sub>3</sub>]<sup>2</sup>/([Br<sub>2</sub>] × [Cl<sub>2</sub>]<sup>3</sup>) E) K<sub>c</sub> = 2[BrCl<sub>3</sub>]<sup>2</sup>/([Br<sub>2</sub>] × 3[Cl<sub>2</sub>]<sup>3</sup>)](https://d2lvgg3v3hfg70.cloudfront.net/TB6485/11eab46b_9ca2_b357_a889_f9541a82d59a_TB6485_11.jpg) Br2(g) + 3Cl2(g)
Br2(g) + 3Cl2(g)
A) Kc = [Br2] [Cl2]/[BrCl3]
B) Kc = [Br2] [Cl2]5/[BrCl3]2
C) Kc = [Br2] [Cl2]3/[BrCl3]2
D) Kc = [BrCl3]2/([Br2] × [Cl2]3)
E) Kc = 2[BrCl3]2/([Br2] × 3[Cl2]3)
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
When the concentrations of both the reactants and products remain constant because the forward and reverse reactions are occurring at the same rate then the system is ________________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If,in a particular process,reactants are able to form products,and products are also able to form reactants,then this process may be described as
A) a reversible process.
B) an elementary process.
C) at equilibrium.
D) forbidden.
E) a forward process.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the equilibrium reaction: N2O4(g)  2NO2(g) Which of the following correctly describes the relationship between Kc and KP for the reaction?
2NO2(g) Which of the following correctly describes the relationship between Kc and KP for the reaction?
A) KP = Kc
B) KP = RT × Kc
C) KP = (RT × Kc ) -1
D) KP = Kc/RT
E) KP = RT/Kc
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the reversible reaction: 2NO2(g)  N2O4(g) If the concentrations of both NO2 and N2O4 are each 0.016 M,what is the value of Qc?
N2O4(g) If the concentrations of both NO2 and N2O4 are each 0.016 M,what is the value of Qc?
A) 0.016
B) 0.50
C) 1.0
D) 2.0
E) 63
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At 250ºC,the equilibrium constant,KP,for the reaction PCl5(g)  PCl3(g) + Cl2(g) is 1.80. Sufficient PCl5 is put into a reaction vessel to give an initial pressure of 2.74 atm at 250ºC.What is the partial pressure of PCl5 after the system has reached equilibrium.
PCl3(g) + Cl2(g) is 1.80. Sufficient PCl5 is put into a reaction vessel to give an initial pressure of 2.74 atm at 250ºC.What is the partial pressure of PCl5 after the system has reached equilibrium.
A) 1.50 atm
B) 1.24 atm
C) 4.24 atm
D) 0.94 atm
E) 1.12 atm
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Suppose 50.0 g of N2O4 is introduced into an evacuated 2.00-L vessel and allowed to come to equilibrium with its decomposition product,N2O4(g)  2NO2(g) .For this reaction Kc = 0.133.Once the system has reached equilibrium,5.00 g of NO2 is injected into the vessel,and the system is allowed to equilibrate once again.What is the mass of NO2 in the final equilibrium mixture?
2NO2(g) .For this reaction Kc = 0.133.Once the system has reached equilibrium,5.00 g of NO2 is injected into the vessel,and the system is allowed to equilibrate once again.What is the mass of NO2 in the final equilibrium mixture?
A) 7.8 g
B) 12.4 g
C) 14.7 g
D) 19.7 g
E) 15.5 g
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The equilibrium constant KP at 427°C for the reaction N2(g) + 3H2(g)  2NH3(g) is 9.4 ×10-5.What is ΔG° for the reaction under these conditions? (R = 8.314 J/K • mol)
2NH3(g) is 9.4 ×10-5.What is ΔG° for the reaction under these conditions? (R = 8.314 J/K • mol)
A) -33 kJ/mol
B) -54 kJ/mol
C) 54 kJ/mol
D) 33 kJ/mol
E) 1.3 J/mol
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrogen sulfide can be formed in the following reaction: H2(g) + S2(g)  H2S(g) ; ΔH°rxn = -92 kJ/mol
The equilibrium constant,KP,is 106 at 1023 K.Estimate the value of KP at 1218 K.(R = 8.314 J/K • mol)
H2S(g) ; ΔH°rxn = -92 kJ/mol
The equilibrium constant,KP,is 106 at 1023 K.Estimate the value of KP at 1218 K.(R = 8.314 J/K • mol)
A) 5.05
B) 18.8
C) 34.7
D) 88.9
E) 598
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Suppose 75.0 g of PCl5(g) is introduced into an evacuated 3.00-L vessel and allowed to reach equilibrium at 250ºC. PCl5(g)  PCl3(g) + Cl2(g)
If KP = 1.80 for this reaction,what is the total pressure inside the vessel at equilibrium? (R =0.0821 atm • L/mol • K)
PCl3(g) + Cl2(g)
If KP = 1.80 for this reaction,what is the total pressure inside the vessel at equilibrium? (R =0.0821 atm • L/mol • K)
A) 2.88 atm
B) 2.28 atm
C) 4.54 atm
D) 7.43 atm
E) 9.69 atm
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which substances are included in the equilibrium constant expression,Kc?
A) Only pure solids
B) Only pure liquids
C) Only pure solids and liquids
D) Only gases and dissolved substances
E) All participating substances
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the gas phase,formic acid forms a dimer,2HCOOH(g)  (HCOOH) 2(g) .For this reaction,ΔH° = -60.1 kJ/mol and ΔG° = -13.9 kJ/mol at 25°C.Find the equilibrium constant (KP) for this reaction at 75 °C.
(HCOOH) 2(g) .For this reaction,ΔH° = -60.1 kJ/mol and ΔG° = -13.9 kJ/mol at 25°C.Find the equilibrium constant (KP) for this reaction at 75 °C.
A) 8960
B) 273
C) 0.120
D) 8.33
E) 1.12 × 10-4
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
If the system 3H2(g)+ N2(g)
 2NH3(g)is at equilibrium and more N2 is added,a net reaction that consumes some of the added N2 will occur until a new equilibrium is reached.
2NH3(g)is at equilibrium and more N2 is added,a net reaction that consumes some of the added N2 will occur until a new equilibrium is reached.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement is correct?
A) When Q < K then ΔG = 1.
B) When Q < K then ΔG = -ΔS.
C) When Q = K then ΔG = 0.
D) When Q > K then ΔG = 1.
E) When Q > K then ΔG = - RT.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A mixture of 0.500 mole of carbon monoxide and 0.400 mole of bromine was placed into a rigid 1.00-L container and the system was allowed to come to equilibrium.The equilibrium concentration of COBr2 was 0.233 M.What is Kc for this reaction?
CO(g) + Br2(g)  COBr2(g)
COBr2(g)
A) 5.23
B) 2.14
C) 1.17
D) 0.467
E) 0.191
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 135
Related Exams