B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based on the phase diagram of a pure substance given below,which statement is true? 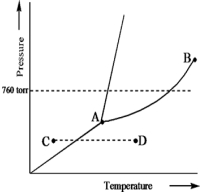
A) The solid phase has a lower density than the liquid phase.
B) The triple point is at a higher temperature than the normal melting point.
C) The substance changes from a solid to a liquid as one follows the line from C to D.
D) The substance changes from a liquid to a gas as one follows the line from C to D.
E) Point B represents the critical temperature and pressure for the substance.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Iron crystallizes in the body-centered cubic lattice.What is the coordination number for Fe?
A) 4
B) 6
C) 8
D) 10
E) 12
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below is the phase diagram for sulfur.Which phase is present at 100°C and 1 atm? 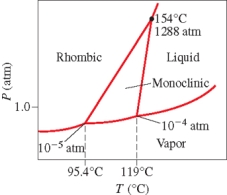
A) liquid
B) vapor
C) rhombic solid
D) monoclinic solid
E) liquid-vapor boundary line
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based on the phase diagram of a pure substance given below,what phase exists at point F? 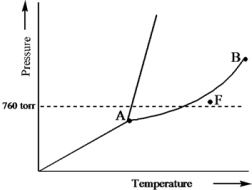
A) vapor + liquid
B) vapor
C) liquid
D) solid
E) supercritical fluid
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What mass of water would need to evaporate from your skin in order to dissipate 1.70 ×105 J of heat from the surface of your body? H2O(l) → H2O(g) ΔHvap = 40.7 kJ/mol
A) 2.26 g
B) 4.18 g
C) 75.2 g
D) 4.18 ×103 g
E) 4.07 × 104 g
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the attractive force between like molecules involved in capillary action?
A) Surface tension
B) Adhesion
C) Polarity
D) Viscosity
E) Cohesion
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is not a valid phase diagram for a pure substance?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The strongest intermolecular interactions between pentane (C5H12) molecules arise from
A) dipole-dipole forces.
B) London dispersion forces.
C) hydrogen bonding.
D) ion-dipole interactions.
E) carbon-carbon bonds.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the pair of substances in which the one with the lower vapor pressure at a given temperature is listed first.
A) ![]()
B) PH3,NH3
C) CF4,CBr4
D) C3H8,C4H10
E) F2,Cl2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of atoms in a face-centered cubic unit cell is
A) 1.
B) 2.
C) 3.
D) 4.
E) 8.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Liquid ammonia can be used as a refrigerant and heat transfer fluid.How much energy is required to heat 25.0 g of NH3(l) from -65.0°C to -12.0°C? Normal boiling point -33.4°C Specific heat of NH3(l) 4.7 J/g • °C Specific heat of NH3(g) 2.2 J/g • °C Molar heat of vaporization 23.5 kJ/mol
A) 4.92 kJ
B) 16.0 kJ
C) 34.5kJ
D) 39.4 kJ
E) 182.8kJ
G) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
______________ is the name given to the phase change from a gas directly to a liquid.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the intermolecular force that exists between a calcium ion and water?
A) dipole-dipole forces
B) London dispersion forces
C) ion-dipole forces
D) covalent bonding
E) ion-ion forces
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A liquid boils when its
A) vapor pressure is exactly 1 atmosphere.
B) vapor pressure is equal to or greater than the external pressure.
C) temperature is equal to 273 K (standard temperature) .
D) temperature is greater than room temperature.
E) thermal energy is equal to the heat of vaporization.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What name is given to the curved surface of a liquid contained in a narrow tube?
A) Surface tension
B) Adhesion point
C) Meniscus
D) Capillary point
E) Cohesion point
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which substance should exhibit hydrogen bonding in the liquid phase?
A) PH3
B) He
C) H2S
D) CH4
E) CH3OH
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the energy in kJ/mol required to melt 1 mole of a solid?
A) molar heat of freezing
B) molar heat of fission
C) molar heat of vaporization
D) molar heat of fusion
E) molar heat of condensation
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following properties indicates the presence of strong intermolecular forces in a liquid?
A) a low heat of vaporization
B) a low critical temperature
C) a low vapor pressure
D) a low boiling point
E) a low melting point
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The strongest intermolecular interactions between hydrogen sulfide (H2S) molecules arise from
A) dipole-dipole forces.
B) London dispersion forces.
C) hydrogen bonding.
D) ion-dipole interactions.
E) disulfide linkages.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 135
Related Exams